 |
Simultaneously detect all major types of structural variants and CNVs that could affect normal function our cause malignancies, on a genome-wide scale. | |
| Ensure the genomic integrity of iPSCs and other cell types. | ||
 |
Reduce turnaround time and cost by characterizing the breadth of SVs and CNVs in a single, scalable NGS-based assay. |
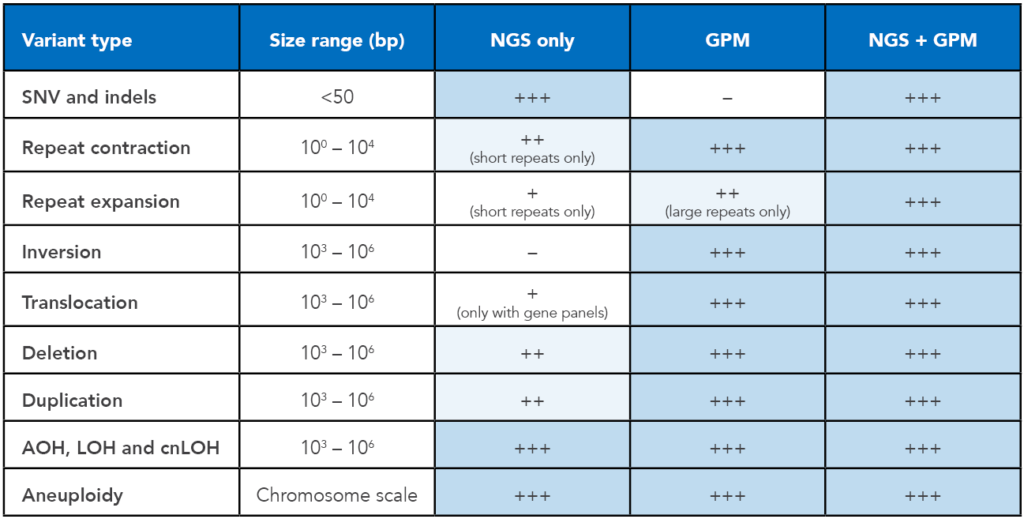
Get the complete picture with NGS and GPM
| Automated analytics yield actionable results
The CytoTerra® Platform provides a sample-to-report NGS-based cytogenetics assay that starts with a small number of cells from your cell therapy development process, and ends with a comprehensive and actionable report in standard and sequence-based nomenclature. Fully automated, cloud-based analysis of paired-end, short-read sequencing data is performed with Phase Genomics’ proprietary computational tools. The entire process can be completed in less than a week, and is scalable to large numbers of samples. |
 |
