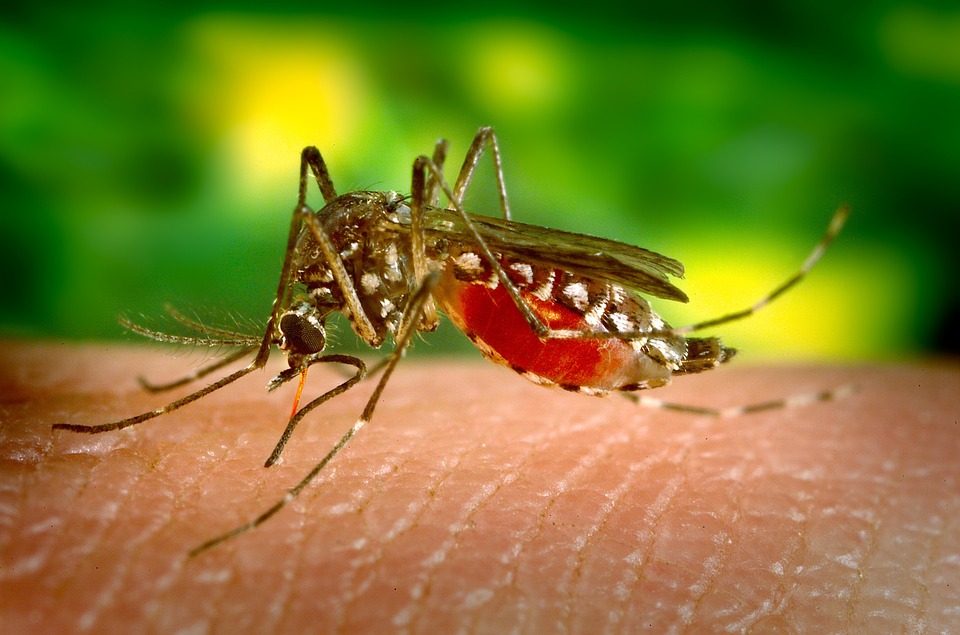
New Genome Published for Malaria Vector Mosquito, An. funestus.
Plasmodium parasites—the microbes that cause malaria—are right at home in the tropics. After all, tropical regions harbor the two animals that the malaria parasites need to complete their complex lifecycle: female Anopheles mosquitoes and human beings. And in 2017 alone, Plasmodiumracked up 219 million cases of malaria, with 435,000 deaths…
Read the full article in Genes to Genomes!
By generating a high-quality genome assembly for one of these mosquitos, researchers in the future will be able to reveal genomic clues as to why An. funestus is able to be a key vector for malaria. Here is our the Q&A with Dr. Nora Besansky, a leading author behind the new genome assembly for malaria vector mosquito, An. funestus.
Why is it important to understand mosquito genetics in malaria research?
Dr. Besansky: Malaria parasites are not spread directly between humans, unlike cold or flu viruses. Malaria mosquitoes — the subset of all mosquitoes that spread the disease — are essential for malaria transmission. Mosquitoes must bite humans to spread the disease, but they don’t operate like dirty syringes. Malaria parasites enter the mosquito when it bites an infected human. The parasite then has to complete a long (at least 10-day) and exquisitely complex developmental process inside the mosquito before that mosquito can infect another human through its bite. The propensity of a mosquito to bite humans, and the ability of the malaria parasite to develop successfully inside of the mosquito depends on mosquito and parasite genetics. Mechanistic understanding of mosquito genetics provides novel opportunities for us to control disease transmission by mosquitoes, without harming the environment or other organisms.
How does access to health care affect treatment for malaria?
Dr. Besansky: Malaria is actually a curable disease — if humans are infected with malaria parasites that are susceptible to the current drugs, and if the infected humans have access to those drugs and to health care. But malaria is a disease of poverty. Health care is not often accessible or affordable, and malaria parasites are rapidly becoming resistant to anti-parasite drugs. Since these drugs have only a short-term effect in the human body, they are impractical for control of malaria in high-transmission parts of the world, even in the absence of parasite resistance, due to the economic burden and the lack of infrastructure for drug distribution.
How does the rise of insecticide use affect the spread of malaria?
Dr. Besansky: In the absence of an effective malaria vaccine, broad-spectrum insecticides against malaria mosquitoes are the mainstay of malaria control. But like malaria parasites, the mosquitoes are also becoming resistant to the insecticides that have been approved for use inside homes and on bed nets, particularly in the face of massively scaled-up insecticide-use campaigns to control malaria. Resistance not only means that mosquitoes can survive exposure to the insecticide; resistance can also be behavioral. For example, mosquitoes that normally enter houses to bite at night, when people are sleeping under bed nets, and rest on indoor walls may change their behavior to daytime biting and outdoor resting — making it much more difficult to specifically target those mosquitoes. Genetic research offers the opportunity to understand in detail aspects of mosquito behavior and physiology that are essential to the mosquito life cycle or to the parasite developmental process inside the mosquito, revealing new and specific ways to intervene and protect human health.
Why is it important to have a chromosome-scale genome assembly for Anopheles funestus?
Dr. Besansky: Human malaria is prevalent in many tropical regions across the globe. But Africa suffers disproportionately. About 90 percent of malaria cases and malaria deaths occur in tropical Africa south of the Sahara. This is mainly due to the dominance of two highly efficient mosquito vectors of human malaria that occur throughout that region: Anopheles gambiae and Anopheles funestus. Owing to its acknowledged importance in malaria transmission, An. gambiae was the first insect, after the fruit fly Drosophila melanogaster, to have its genome fully sequenced and assembled. Sequencing and assembly of other anopheline malaria vectors has lagged, but in 2015, an additional 16 Anopheles reference genomes were made available, among these An. funestus. However, limitations of the sequencing technologies applied at that time meant that these reference genomes were not chromosome-scale assemblies. Just as driving from New York to Los Angeles is facilitated by a road map, genetic research also is more powerful, efficient and accurate if the 260 million puzzle pieces of nucleotides in the nuclear genome are properly ordered and oriented.
Does heterozygosity or haplotype diversity affect genome assembly methods for the Anopheles species?
Dr. Besansky: A major international consortium, modeled after the 1,000 human genomes consortium, published its findings based on 765 An. gambiae mosquitoes sampled from natural populations. A major conclusion was that, on average, there is a polymorphic site every other nucleotide in An. gambiae, emphasizing the almost unprecedented heterozygosity of this species. This same consortium has begun work on An. funestus, a species expected to be equally diverse. Such nucleotide diversity is well-known to pose difficulties for chromosome-scale assemblies based on traditional sequencing technologies.
What are chromosomal inversions? Do they affect the spread of malaria?
Dr. Besansky: Chromosomal inversions are reversals in gene order that occur when a linear chromosome breaks in two places, and the intervening segment rotates 180 degrees before rejoining the other two pieces. They affect the spread of malaria, indirectly if not directly, because they typically contain hundreds or thousands of genes involved in climatic or local adaptation, allowing their mosquito carriers to fully exploit heterogeneous and otherwise challenging environments. Due to their modification of recombination rates along the chromosome, undetected inversions can mislead genome-wide association studies and other genetic studies. Chromosome-scale assemblies make it possible to localize inversions in the genome.
