Antibiotic resistance is a rapidly growing global health threat as bacteria share and spread resistance genes via plasmids and other mobile genetic elements. Several teams of researchers applied a new method to understand which microorganisms house genes for antibiotic resistance within complex microbiome communities.
Read the paper, Linking the Resistome and Plasmidome to the Microbiome.
ANTIMICROBIAL RESISTANCE ON THE RISE
According to the World Health Organization, antimicrobial resistance (AMR) in microbial pathogens is expected to take 10 million lives by 2050 if there are no new pharmaceutical or technological advancements dedicated to combating this pressing problem. For almost a century, medicine has made remarkable impact on human life by using antibiotics to treat infections, but this has led to a very concerning overuse problem, stoking an arms race between antibiotics and the pathogens they target. The CDC points out that at least 30% of antibiotic prescriptions are unnecessary and there is a massive contribution to antibiotic overuse in the food and agriculture industry where each year 130,000 tons of antibiotics are given to food animal livestock. Both of these problems correlate with the rise of AMR.
Though there are naturally occurring antibiotic-resistant bacteria, there are two mechanisms by which bacteria can acquire antimicrobial resistance genes (ARGs) and become resistant: 1) through spontaneous genetic mutations and/or 2) by acquiring genetic material from other microbes via plasmids, viruses, or other means of horizontal gene transfer. Due to the evolutionary pressure exerted on microbes by antibiotic overuse, pathogens resistant to these antibiotics within our body, hospitals, and the environment become reservoirs of transmittable AMR genes that can rapidly spread and accumulate within a single microbe contributing to the emergence of multidrug-resistant microbes commonly known as superbugs.
PROXIMITY-LIGATION (HI-C) LINKS ARG AND PLASMIDS TO THEIR HOSTS
One of the biggest obstacles faced by scientists when studying AMR is the inability to determine which microbes are carrying and spreading specific ARGs. Because these genes often travel on mobile elements, they can move dynamically between different species and can therefore be found in numerous organisms without one clear parental host. When attempting to sequence the DNA of a mixed microbial sample, all the DNA is purified from all the cells at the same time and the host-plasmid connection is severed, making it nearly impossible to determine where each mobile element came from or if they were shared among several species. In this newly published paper, researchers highlight a novel method for linking ARGs and other mobile genetic elements to their hosts directly from microbiome samples using the latest version of the proximity-ligation (Hi-C) data analysis tool, ProxiMeta Hi-C.
Phase Genomics CEO, Dr. Ivan Liachko, describes how our Hi-C platform solves one of microbiologists’ greatest problems pertaining to the linking of plasmids with their hosts.
Hi-C utilizes in vivo proximity-ligation which can assemble complete genomes down to the strain-level directly from mixed-population samples as well as physically links plasmids/ARGs to their host. This method is particularly useful for researchers studying the “dark-matter” of the microbiome because the method does not require culturing nor a priori information about a sample.
USING HI-C TO TRACK ARGs IN THE MICROBIOME
Lead author Thibault Stalder from the University of Idaho used the ProxiMeta Hi-C kit on a complex microbiome wastewater community, a suspected AMR reservoir, to learn more about which bacteria carry ARGs. After the Hi-C library was sequenced, Phase Genomics used the data to inform contig clustering of hundreds of genomes, most of which are novel, with our cloud-based software – ProxiMeta. Using the genome clusters found by ProxiMeta, the Hi-C linkages of each ARG-, plasmid-, and integron-bearing contigs to each genome were measured to determine which species physically hosted the relevant mobile elements.
ProxiMeta was able to cluster contigs into >1000 genome clusters and search for over 30 groups of ARGs, plasmids, and integrons which speed up the adaptive process of newly integrated ARGs (Figure 1, circle plot). For each of these genes, we inferred hosts (Figure 2). Moreover, these organisms generally belonged to families known to host each known gene (marked with an “X” in Figure 2), supporting the accuracy of the analysis. In the future, this information will allow us to track the spread of AMR in complex communities consisting of many diverse organisms.
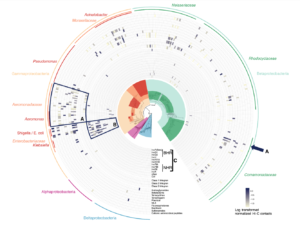
Figure 1: Hi-C linkage between ARGs, plasmid markers, and integrons among clusters belonging to Alpha, Beta, Gamma and Delta Proteobacteria.
Over 200 genome clusters had strong Hi-C links to ARGs, of which 12 had high-quality assemblies. These resultant genomes include both gram positive and gram-negative bacteria and most belonged to species that were previously unsequenced. ARGs were mostly linked to genome clusters belonging to the Gammaproteobacteria, Betaproteobacteria and Bacteroidetes (Figure 2, below).
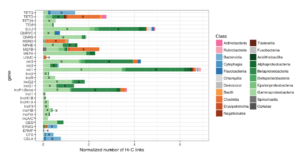
Figure 2: Normalized Hi-C links between ARGs, plasmids, and families of bacteria.
FUTURE DIRECTIONS
This method can be useful for researchers not only studying the microbiome, but the virome as well. Phages, or viruses, also distribute genetic information amongst bacteria to influence host biology, much like plasmids. Several previous studies showed that in vivo proximity-ligation can be used to link phages with their hosts directly from mixed complex samples, much like was done with plasmids and AMR genes in this study. This information could be crucial to labs and companies that are now engineering phages that could replace the widespread use of antibiotics and combat AMR.
This year, antibiotic resistant bugs have infected more than 2 million people globally; 23,000 of those individuals will die because of our inability to fight these superbugs. By using ProxiMeta Hi-C to better understand the genomics of microbial communities suspected to be AMR reservoirs, researchers can identify ARG carriers down to the strain-level and quantify how prevalent these genes are. With further exploration, this tool could one day offer a new solution to limit the spread of these genes and reverse the trend of increasing antibiotic resistance and save lives.
BRING A HI-C KIT INTO YOUR LAB TODAY
Phase Genomics offers a wide variety of proximity-ligation products and services including Hi-C preparation kits and a range of different cloud-based bioinformatic analysis platforms. Power your microbiome research with ProxiMeta Hi-C and our easy Hi-C kits; assemble hundreds of complete genomes for novel, unculturable microbes, and associate plasmids with hosts directly from raw microbiome samples using ProxiMeta Hi-C.

 Twitter
Twitter LinkedIn
LinkedIn Email
Email