Congratulations to Dr. Ben Tully on winning this year’s Project ProxiMeta: 2019 Metagenomics Award! Read more about his project, 4. The Complete Hydrothermal Microbial Metal Metabolism
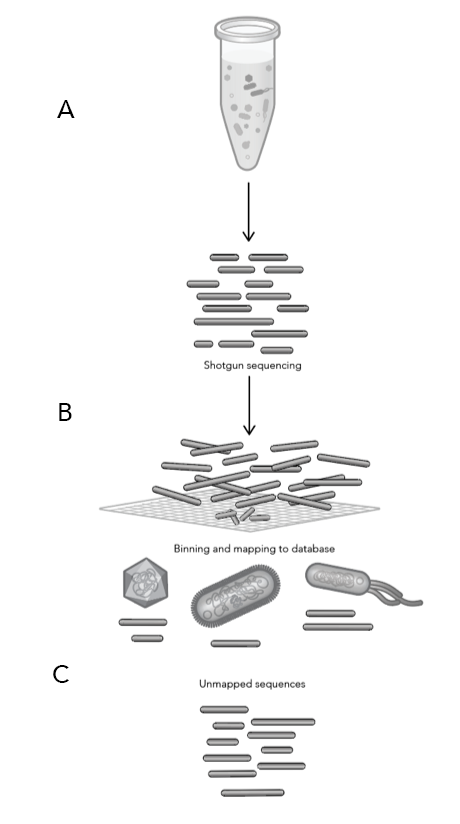
This summer, researchers from across the U.S. sent in short proposals for a chance to win a full-service ProxiMeta™ microbiome workup for a sample of their choice. ProxiMeta combines shotgun metagenomics with in vivo proximity ligation (Hi-C) and necessary bioinformatic tools to help researchers assemble high-quality microbial genomes directly from complex microbiome samples.



HOW TO VOTE
Each project was assessed by a panel of scientists for scientific merit, novelty, impact, and feasibility, and four finalists were selected. Cast your vote on Twitter for your favorite project.
THE FINALISTS
1. The Gut Microbiome as a Risk Factor for Arsenic-Induced Cancer
Twitter Name: Gut & As-Induced Cancer
 It is estimated that ~200 million people worldwide are exposed to arsenic concentrations exceeding current safety standards. Our collaborators have recently demonstrated that mice and human microbiomes can protect mice from arsenic toxicity. While human stool supplementation fully restores protection to arsenic in germ-free mice, researchers were only able to isolate one microbe, Faecalibacterium prausnitzii, that successfully conferred protection to both parent and infant mice. These results are huge because arsenic poses the highest lifetime risk for developing cancer in humans.We will investigate the role of arsenic-transforming bacteria within the gastrointestinal (GI) microbiome as another possible risk factor.
It is estimated that ~200 million people worldwide are exposed to arsenic concentrations exceeding current safety standards. Our collaborators have recently demonstrated that mice and human microbiomes can protect mice from arsenic toxicity. While human stool supplementation fully restores protection to arsenic in germ-free mice, researchers were only able to isolate one microbe, Faecalibacterium prausnitzii, that successfully conferred protection to both parent and infant mice. These results are huge because arsenic poses the highest lifetime risk for developing cancer in humans.We will investigate the role of arsenic-transforming bacteria within the gastrointestinal (GI) microbiome as another possible risk factor.
In nature, arsenic-reducing microorganisms are well known for their ability to generate more toxic arsenic products called arsenites, which are typically formed in anaerobic environments like the gut. Past research indicates that ingested arsenic may also be transformed into the toxic product arsenite by gut microbes thus increasing the risk for the host. On the other hand, arsenite-oxidizing microbes may also provide a benefit to the host by lowering arsenite concentrations. The ability of the microbiome to transform arsenic is determined by its genetic composition, therefore ProxiMeta sequencing technology will allow us to immediately analyze our collaborators rodent stool samples for genetic clues regarding this mysterious protection. Our project goals are to expand on this knowledge by: (1) characterizing the genetic basis for protection to arsenic provided by the microbiome (2) identifying, and then isolating, the bacteria-harboring arsenic transforming genes involved in protection.
We predict that differences in the gut metagenome composition will explain the incidences in arsenic susceptibility within a population or even at the family level. This project will provide important insight regarding how gut microbes contribute to cancer and may lead to novel therapies and probiotics that could target the microbiome of arsenic-exposed individuals.
2. Evaluating Antimicrobial Resistance in Backyard Poultry Environments
Twitter: AMR in Backyard Poultry
 Approximately 13 million rural, urban, and suburban US residents reported owning backyard poultry (BYP) in 2014, and interest in BYP ownership is nearly four times that amount. BYP ownership has risen recently due to product quality, public health, ethical, and animal welfare concerns of commercial operations. However, BYP ownership and disease treatment is largely under-regulated, unlike commercial poultry production. Lack of regulation poses public health concerns of transmission of antimicrobial resistant (AMR) bacteria, such as AMR strains of Salmonella, Mycoplasma gallisepticum, and Escherichia coli commonly associated with BYP. BYP owners (2014 survey) were largely uninformed about poultry diseases and treatments but were interested in learning more on disease management.
Approximately 13 million rural, urban, and suburban US residents reported owning backyard poultry (BYP) in 2014, and interest in BYP ownership is nearly four times that amount. BYP ownership has risen recently due to product quality, public health, ethical, and animal welfare concerns of commercial operations. However, BYP ownership and disease treatment is largely under-regulated, unlike commercial poultry production. Lack of regulation poses public health concerns of transmission of antimicrobial resistant (AMR) bacteria, such as AMR strains of Salmonella, Mycoplasma gallisepticum, and Escherichia coli commonly associated with BYP. BYP owners (2014 survey) were largely uninformed about poultry diseases and treatments but were interested in learning more on disease management.
The combination of a lack of regulation and public information warrants further research into the bacterial communities of BYP and their environments. Cloacal and environmental swabs were collected as part of a 2018 citizen science study where BYP owners reported current and historical poultry antibiotic usage. We propose to conduct shotgun metagenomic sequencing and proximity ligation using the ProxiMeta platform, allowing for increased detection of full-length AMR gene alleles compared to that revealed by short-read sequencing. The combination of PacBio reads with HiC intercontig ligation analysis allows for identification of potential gene transfer events of AMR genes within communities and potential dissemination throughout the environment.
This analysis is especially important considering the public health concerns of AMR persistence in backyard environments. Additionally, investigation of lytic and prophage presence would allow investigation of phage-mediated bacterial regulation that would not be possible with short-read sequencing alone. ProxiMeta analysis of these samples would provide the most comprehensive insight of AMR presences and persistence in BYP environments to date. These findings will be critical for new regulation and disease management for the increasing number of BYP flocks, which currently pose a potential health risk.
3. Unraveling the Metagenomics of Contamination
Twitter: Steel Site Contamination
 We propose a metagenome characterization of contaminated Munger Landing sediment located in the St. Louis River, Duluth, MN USA. Seasonal samples are already collected and stored; of which one will be sequenced. Munger landing, is located downstream from the U.S. Steel Superfund site and contaminants include PAHs, dioxins, PCBs, and heavy metals.
We propose a metagenome characterization of contaminated Munger Landing sediment located in the St. Louis River, Duluth, MN USA. Seasonal samples are already collected and stored; of which one will be sequenced. Munger landing, is located downstream from the U.S. Steel Superfund site and contaminants include PAHs, dioxins, PCBs, and heavy metals.
Soil condition is integral to high productivity and ecosystem balance at all trophic levels. Human activities erode soil condition through agriculture, mining, sewage outflows and/or chemical/waste disposal into waterways. These practices alter the chemical structure of the soil and break down the microbial community processes responsible for ensuring the balance of biogeochemical cycling patterns in the soil. We hypothesize the activity of these pathways involved in cycling of nitrogen, phosphorus and carbon are altered in contaminated soil systems.
Metagenomic profiling of Munger Landing will provide data to examine microbes, metabolic pathways, and contaminant-processing genes present in the community that can be characterized further using qRTPCR. This project will be presented within a community college microbiology course module. Curriculum utilizing real-world data and the sequencing technology from Phase Genomics will teach students experimental design, troubleshooting, hypothesis testing, data analysis and how to communicate the broader impacts of a study to society, the field of environmental microbiology or conservation.
In the future, this data will assist in designing a longitudinal metagenomic and metatranscriptomic study to assess the ability of remediation to ‘recover’ bacterial community function at the Munger Landing site; slated to start in 2020-2021 as compared to two uncontaminated control sites. Ten sites, slated for remediation, have been identified as having high chemical and heavy metal contamination for the St. Louis River Estuary. The Munger Landing project will establish a workflow that can be applied to other contaminated sites.
4. The Complete Hydrothermal Microbial Metal Metabolism
Twitter: Hydrothermal Microbiome
 Hydrothermal vents replenish the oceans with much-needed micronutrients, spewing iron, magnesium, nickel, and other metals from the earth’s crust. These metal micronutrients are used as biological cofactors for organisms throughout the marine food chain. Boiling, sterile hydrothermal fluids quickly cool and are colonized by highly specialized microorganisms that begin to cycle the metal species mixing with the seawater. Though regularly sampled, rarely have hydrothermal plumes been tracked through the water column to establish how microbial colonization occurs through time and space. We lack understanding regarding the replicability of colonization to what extent stochastic processes shape microbial community structure.
Hydrothermal vents replenish the oceans with much-needed micronutrients, spewing iron, magnesium, nickel, and other metals from the earth’s crust. These metal micronutrients are used as biological cofactors for organisms throughout the marine food chain. Boiling, sterile hydrothermal fluids quickly cool and are colonized by highly specialized microorganisms that begin to cycle the metal species mixing with the seawater. Though regularly sampled, rarely have hydrothermal plumes been tracked through the water column to establish how microbial colonization occurs through time and space. We lack understanding regarding the replicability of colonization to what extent stochastic processes shape microbial community structure.
While on station at the East Pacific Rise hydrothermal vent field, size-fractionated samples (0.2, 3.0 and 5.0-μm) were collected in the hydrothermal plume emanating from Bio Vent. Samples fluids were collected from the source through the first 1-km of dispersal – the key distance for colonization – and this effort was repeated over the course of 10-days – to determine the replicability of natural colonization events. The application of standard metagenomics sequencing and microbial genome reconstruction through binning would provide novel insight into the cycling of metals within the plume but the use of cross-linked DNA techniques would deliver an unprecedented understanding of how strain diversity impacts colonization and how microbes interact with extrachromosomal elements in the environment.
While some microbes are poised to take advantage of reduced metal species for lithotrophic growth, microbes from the water column that become entrained in the plume will need metal-resistance adaptations to alleviate stress from the elevated metal concentrations present. Metal-resistance genes dispersed through the viral and plasmid pools are essential elements for understanding the functioning of the microbial community in this globally important source of metals to the oceans and effective interpretation of the community can only be achieved through cross-linked DNA metagenomic techniques.
*All finalists projects are owned by verified researchers at U.S. academic institutions.
RESOURCES
- Watch the Video: How it Works: ProxiMeta Metagenome Deconvolution
- Paper: Linking the Resistome and Plasmidome to the Microbiome, ISME Journal (May 30, 2019)
- View Example ProxiMeta Reports
- Review Contest Rules
- Standard ProxiMeta Project Guidelines *For Research Purposes Only. Not for Diagnostic or Clinical Use.





















 Twitter
Twitter LinkedIn
LinkedIn Email
Email